|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
HANNOVER FAIR 1995 - 2019
 HANNOVER FAIR 2019 HANNOVER FAIR 2019
 HANNOVER FAIR 2018 HANNOVER FAIR 2018
 HANNOVER FAIR 2017 HANNOVER FAIR 2017
 Arno's Forum Interview 2017 Arno's Forum Interview 2017
 HANNOVER FAIR 2016 HANNOVER FAIR 2016
 HANNOVER FAIR 2015 HANNOVER FAIR 2015
 HANNOVER FAIR 2014 HANNOVER FAIR 2014
 HANNOVER FAIR 2013 HANNOVER FAIR 2013
 HANNOVER FAIR 2012 HANNOVER FAIR 2012
 HANNOVER FAIR 2011 HANNOVER FAIR 2011
 HANNOVER FAIR 2010 HANNOVER FAIR 2010
 Arno's
Forum Interview 2010 Arno's
Forum Interview 2010
 HANNOVER FAIR 2009 HANNOVER FAIR 2009
 Arno's Video during HF 2009 Arno's Video during HF 2009
 Arno's Forum Interview 2009 Arno's Forum Interview 2009
 HANNOVER FAIR 2008 HANNOVER FAIR 2008
 Arno's Forum Interview 2008 Arno's Forum Interview 2008
 HANNOVER FAIR 2007 HANNOVER FAIR 2007
 Arno's Forum Interview 2007 Arno's Forum Interview 2007
 HANNOVER FAIR 2006 HANNOVER FAIR 2006
 Daily Networking evenings Daily Networking evenings
 HANNOVER FAIR 2005 HANNOVER FAIR 2005
 Daily Networking evenings Daily Networking evenings
 International
Commercial Visitors 2005 International
Commercial Visitors 2005
 HANNOVER FAIR 2004 HANNOVER FAIR 2004
 International
Commercial Visitors 2004 International
Commercial Visitors 2004
 HANNOVER FAIR 2003 HANNOVER FAIR 2003
 International
Commercial Visitors 2003 International
Commercial Visitors 2003
 HANNOVER FAIR 2002 HANNOVER FAIR 2002
 HANNOVER FAIR 2001 HANNOVER FAIR 2001
 HANNOVER FAIR 2000 HANNOVER FAIR 2000
 HANNOVER FAIR 1999 HANNOVER FAIR 1999
 HANNOVER FAIR 1998 HANNOVER FAIR 1998
 HANNOVER FAIR 1997 HANNOVER FAIR 1997
 HANNOVER FAIR 1996 HANNOVER FAIR 1996
 HANNOVER FAIR 1995 HANNOVER FAIR 1995
|
| |
| Visits and Workshops |
 |
 Hans-Olof Nilsson's Off-The-Grid Hydrogen House near Goteborg, Sweden Hans-Olof Nilsson's Off-The-Grid Hydrogen House near Goteborg, Sweden |
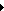 |
 Visit at the Italian demonstration plant of MagneGas⢠on February 7th and 8th, 2011 Visit at the Italian demonstration plant of MagneGas⢠on February 7th and 8th, 2011 |
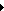 |
 Hydrogen Plant of Emirates Industrial Gases Co. Ltd ( EIGC) at Dubai, UAE Hydrogen Plant of Emirates Industrial Gases Co. Ltd ( EIGC) at Dubai, UAE |
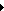 |
 Huerta Solar en Tabernas, Spain, October 2009 Huerta Solar en Tabernas, Spain, October 2009 |
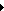 |
 Andasol, Spain, October 2009 Andasol, Spain, October 2009 |
 |
 Antares DLR H2, Stuttgart, Germany,
September 2009 Antares DLR H2, Stuttgart, Germany,
September 2009 |
 |
 German Aerospace Center (DLR) Deutsches Zentrum für Luft-und Raumfahrt eV, Stuttgart, Germany, Institute of Technical Thermodynamics (ITT) Institute of Vehicle Concepts Stuttgart, Germany, June 2009 German Aerospace Center (DLR) Deutsches Zentrum für Luft-und Raumfahrt eV, Stuttgart, Germany, Institute of Technical Thermodynamics (ITT) Institute of Vehicle Concepts Stuttgart, Germany, June 2009 |
 |
 Brennstoffzellen-Boote für den Freizeitbereich, Hochschule Konstanz, Konstanz, Germany June 2009 Brennstoffzellen-Boote für den Freizeitbereich, Hochschule Konstanz, Konstanz, Germany June 2009 |
 |
 AFCC Automotive Fuel Cell Cooperation, Burnaby, BC, Canada June 2009 AFCC Automotive Fuel Cell Cooperation, Burnaby, BC, Canada June 2009 |
 |
 Powertech Labs Inc., a: "... wholly owned subsidiary of BC Hydro (a Crown corporation of the Government of British Columbia), Surrey, BC, Canada June 2009 Powertech Labs Inc., a: "... wholly owned subsidiary of BC Hydro (a Crown corporation of the Government of British Columbia), Surrey, BC, Canada June 2009 |
 |
 Plataforma Solar de Almería, Spain Plataforma Solar de Almería, Spain |
|
| Observations |
 |
 How Airbus conquered the How Airbus conquered the
US market in the 70s |
 |
 The Arecibo Observatory The Arecibo Observatory
Arecibo, Puerto Rico |
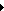 |
 Hat Creek Radio Observatory, Hat Creek Radio Observatory,
Hat Creek, CA, USA |
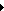 |
 National Radio Astronomy Observatory, Green Bank, WV, USA National Radio Astronomy Observatory, Green Bank, WV, USA |
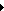 |
 Wright Brothers National Memorial, Kitty Hawk, NC, USA Wright Brothers National Memorial, Kitty Hawk, NC, USA |
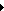 |
 Fuel Cell Bus Trial, Perth, Australia Fuel Cell Bus Trial, Perth, Australia |
 |
 California Hydrogen Highway, USA California Hydrogen Highway, USA |
 |
 SPACEFEST 2009, San Diego, USA SPACEFEST 2009, San Diego, USA |
|
| Impressions from worldwide Conferences, which
we attended to promote the commercialisation of Hydrogen and Fuel
Cells: |
| 2022 |
| #117 |
2nd Edition of International
Conference on
Green Chemistry and Renewable Energy |
| 2020 |
| #116 |
2020 6th International 
Conference on Environment and Renewable Energy (ICERE 2020), 24-26 February 2020, Hanoi, Vietnam |
| 2019 |
| #115 |
4th Annual ASEAN Solar + 
Energy Storage Congress & Expo 2019 14- 15 Nov, 2019 The Bellevue Manila, Philippines |
| #114 |
SFERA-III 1st Summer School &
Doctoral Colloquium at CNRS-PROMES in Odeillo, France
September 9th-13th, 2019 |
| #113 |
Starmus V, June 24 â 29, 2019
Zurich, Switzerland a global festival of science communication and art |
| 2018 |
| #112 |
Visit to NOORo I to III in 
Ouarzazate, Morocco as part of the SolarPACES 2018 conference, October 6, 2018 |
| #111 |
The Hydrogen Energy Summit 
2018 Chiang Mai, Northern Thailand Arno's presentation: Off the Grid â Unveiling new ways for our Energy supply January 26, 2018 |
| 2015 |
| #110 |
1. Klimaschutzkongress auf der 
Insel Sylt
25. September 2015
Vortrag von Arno A. Evers:
Eine âInsel- Lösungâ für Sylt?
Neue Wege zur Energieversorgung der Insel Sylt |
| 2014 |
| #109 |
6. Hamburger Klimawoche 
29. August 2014
Vortrag von Arno A. Evers:
Physikalische und Gesellschaftliche Rahmenbedingungen der Energiewende010 |
| 2012 |
| #108 |
MesseKongress RegioEnergie+++  Dreieich 2012 Dreieich 2012
9. September 2012
Vortrag von Arno A. Evers: Sind wir noch zu retten? |
| 2010 |
| #107 |
WREC World Renewable Energy Congress XI
September 25-30, 2010 
Abu Dhabi, United Arab Emirates
Oct 2010 |
| #106 |
18th World Hydrogen Energy Conference WHEC 2010,
May 16 - 21 
Essen, Germany
Jun 2010 |
| 2009 |
| #105 |
"Bright
Horizons 6" -
A Journey
to the Edge of the Cosmos 
Eastern Caribbean
Dec 2009 |
| #104 |
2009 Fuel Cell Seminar & Exposition
Palm Springs, CA, USA
Nov 2009 |
| #103 |
Fuel Cell Technologies:
FUCETECH 2009
Mumbai (Bombay), India
Nov 2009 |
| #102 |
f-cell
Stuttgart, Germany
Sep 2009 |
| #101 |
SolarPACES 2009
Berlin, Germany
Sep 2009 |
| #100 |
5th Annual Hydrogen Implementation Conference
Charleston, WV, USA
Aug 2009 |
| #99 |
Intersolar North America
San Francisco, CA, USA 
Jul 2009 |
| #98 |
European FUEL CELL FORUM 2009
Lucerne, Switzerland 
Jun 2009 |
| #97 |
HFC2009
Vancouver, Canada 
Jun 2009 |
| #96 |
telescon 2009
Vienna, Austria 
May 2009 |
| #95 |
Hydrogen Works
San Diego, CA, USA 
Feb 2009 |
| #94 |
ICEPAG 2009
Newport Beach, CA, USA 
Feb 2009 |
| 2008 |
| #93 |
HTE-HI.TECH.EXPO 2008
Milan, Italy 
Nov 2008 |
| #92 |
Fuel Cell Seminar & Exposition
Phoenix, AZ, USA 
Oct 2008 |
| #91 |
H2Expo
Hamburg, Germany 
Oct 2008 |
| #90 |
f-cell
Stuttgart, Germany 
Sep 2008 |
| #89 |
INTELEC 2008
San Diego, CA, USA
Sep 2008 |
| #88 |
2008 Formula Zero Championship
Rotterdam, The Netherlands
Aug 2008 |
| #87 |
HyForum 2008
Changsha, P.R. China
Aug 2008 |
| #86 |
WREC X 2008
Glasgow, Scotland, UK
Jul 2008 |
| #85 |
KMCM 2008
Vancouver, BC, Canada
Jul 2008 |
| #84 |
Lucerne FUEL CELL FORUM 2008 
Lucerne, Switzerland
Jun 2008 |
| #83 |
17th World Hydrogen
Energy Conference (WHEC) 
Brisbane, Australia
Jun 2008
 The role of the young generation ... The role of the young generation ... |
| #82 |
Renewable Energy Asia
Bangkok, Thailand 
Jun 2008 |
| #81 |
Selected Hydrogen Fueling Stations in California, USA 
Apr 2008 |
| #80 |
NHA Annual Hydrogen Conference 2008
Sacramento, CA, USA
Mar / Apr 2008 |
| #79 |
FC EXPO 2008 
Tokyo, Japan
Feb 2008 |
| #78 |
Der 4. Deutsche Wasserstoff 
Congress 2008
Essen, Germany
Feb 2008 |
| #77 |
ISEPD 2008 
Changwon, Korea
Jan 2008 |
| 2007 |
| #76 |
20TH World Energy Congress & Exhibition 
Rome, Italy
Nov 2007 |
| #75 |
World Hydrogen Technologies Convention (WHTC) 
Montecatini Terme, Italy
Nov 2007 |
| #74 |
2007 Fuel Cell Seminar & Exposition
San Antonio, Texas, USA
Oct 2007 |
| #73 |
KOREA ENERGY SHOW 2007 
Seoul, Republic of Korea
Oct 2007 |
| #72 |
Tenth Grove Fuel Cell Symposium 
London, UK
Sep 2007 |
| #71 |
Solar Tech India 2007 
New Delhi, India
Sep 2007 |
| #70 |
SES-Fachtagung
MYTHOS STROMLÜCKE 
Zurich, Switzerland
Aug 2007 |
| #69 |
HFCE 2007 
Shanghai, P.R. China
Jul 2007 |
| #68 |
IHEC 2007 
Istanbul, Turkey
Jul 2007 |
| #67 |
KMCM 2007 
Düsseldorf, Germany
Jul 2007 |
| #66 |
Kick Off Meeting zur Leitinnovation Mikrobrennstoffzelle 
Munich, Germany
Jun 2007 |
| #65 |
Hydrogen and Fuel Cells 2007: International Conference &
Trade Show
Vancouver, BC, Canada
April / May 2007 |
| #64 |
GENERA - Energy and Environment International Fair 
Madrid, Spain
Feb / Mar 2007 |
| #63 |
World Renewable Energy Congress [WREN] 
Fremantle, Australia
Feb 2007 |
| #62 |
Environment 2007
Exhibition & Conference 
Abu Dhabi, UAE
Jan 2007 |
| 2006 |
| #61 |
2nd Annual Fuel Cells Durability & Performance 2006 
Miami Beach, FL USA
Dec 2006 |
| #60 |
EDTA Conference & Exposition 
Washington, DC, USA
Nov 2006 |
| #59 |
The Fuel Cell Seminar 
Honolulu, Hawaii, USA
Nov 2006 |
| #58 |
Fraunhofer Symposium
Mikroenergietechnik
POWER TO GO
Berlin, Germany
Oct 2006 |
| #57 |
Renewables to Hydrogen Forum 
Albuquerque, NM, USA
Oct 2006 |
| #56 |
Alternative Transport Energies Conference 
Perth, Western Australia
Sep 2006 |
| #55 |
Power-Gen Asia 
Hong Kong, Hong Kong
Sep 2006 |
| #54 |
World Renewable Energy
Congress IX and Exhibition 
Florence, Italy
Aug 2006 |
| #53 |
R&D in the field of Hydrogen and Fuel Cell in Germany and Europe 
Clausthal, Germany
Jul 2006 |
| #52 |
Lucerne Fuel Cell Forum 2006 
Lucerne, Switzerland
Jul 2006 |
| #51 |
16th World Hydrogen Energy Conference (WHEC) 
Lyon, France
Jun 2006 |
| #50 |
NHA Annual Hydrogen Conference 
Long Beach, CA, USA
Mar
2006 |
| #49 |
FC
EXPO 2006
Tokyo, Japan
Jan 2006 |
| #48 |
Wasserstoff und Brennstoffzellen im Automobil
Essen, Germany
Apr 2006 |
| 2005 |
| #47 |
Fuel
Cell Seminar
Palm Springs, USA
Nov 2005 |
| #46 |
Internationale
ASUE-Fachtagung
Essen, Germany
Nov 2005 |
| #45 |
EHEC
2005
Zaragoza, Spain
Nov 2005 |
| #44 |
Fuel
Cell Summit:
A Road Map to Commercialization
Uncasville, CT, USA
Oct 2005 |
| #43 |
2005
Grove Fuel Cell Symposium
London, UK
Oct. 2005 |
| #42 |
WHTC
2005 World Hydrogen Technologies Convention (WHTC) Singapore, Singapore
Oct. 2005 |
| #41 |
f-cell
2005, Stuttgart, Germany
Sep. 2005 |
| #40 |
The
27th International Telecommunications Energy Conference - intelec
'05
Berlin, Germany
Sep. 2005 |
| #39 |
IHK
Nord Wasserstoff – Tagung
Lübeck, Germany
Sep. 2005 |
| #38 |
ICHS
- International Conference on Hydrogen Safety , Pisa, Italy
Sep. 2005 |
| #37 |
IHEC-2005
International Hydrogen
Energy Congress & Exhibition
Jul. 2005 |
| #36 |
93.
Bunsen Kolloquium
Jun. 2005 |
| #35 |
European
Hydrogen and Fuel Cell Technology Platform (HFP)
Brussels, Belgium
Mar. 2005 |
| #34 |
Cairo
9th International Conference on Energy & Environment
Sharm El-Sheikh, Egypt
Mar. 2005 |
| #33 |
1st
International Fuel Cell Expo
Tokyo, Japan
Jan. 2005 |
| 2004 |
| #32 |
H2PS:
The 2004 Hydrogen Production and Storage Forum,
Washington, DC, USA
Dec. 2004 |
| #31 |
Impressions
from Shanghai
Nov. 2004 |
| #30 |
Renewable Energies China incl. Hydrogen + Fuel Cells
Shanghai, PR China
Nov. 2004 |
| #29 |
Michelin
Challenge Bibendum 2004
Oct. 2004 |
| #28 |
Energy
Asia 2004
Oct. 2004 |
| #27 |
Hydrogen
and Fuel Cells 2004
Conference and Trade Show
Toronto, ON, Canada
Sep. 2004 |
| #26 |
Meetings
in Singapore,
Sep. 2004 |
| #25 |
Hydrogen
and Fuel Cell Futures Conference, Perth, Australia
Sep. 2004 |
| #24 |
Exhibiting
at World Renewable Energy Congress VIII
Denver, CO, USA
Sep. 2004 |
| #23 |
Arno
presenting at ACS National Meeting Philadelphia, PA, USA
Aug. 2004 |
| #22 |
Promotion
of FP6, for European Union, Delegation of the European Commission,
Shanghai
Jul. 2004 |
| #21 |
IHK
Energy-Podium 2004
Jul. 2004 |
| #20 |
15th
World Hydrogen Energy Conference (WHEC15)
Yokohama, Japan
Jun. 2004 |
| #19 |
Impressions
from the
Energy Forum 2004
Varna, Bulgaria
Jun. 2004 |
| #18 |
Impressions
from HYFORUM
May 2004 |
| #17 |
Impressions
from Dubai
United Arab Emirates
May 2004 |
| #16 |
Impressions
from Argentina
May 2004 |
| #15 |
Promoting
Hydrogen Production from Patagonia, Argentina
May 2004 |
| #14 |
Impressions
Zhuozheng Garden
in Su Zhou
Mar. 2004 |
| 2003 |
| #13 |
H2PS:
The 2003 Hydrogen Production and Storage Forum
Washington, D.C., USA
Dec. 2003 |
| #12 |
Impressions
from
Washington, D.C., USA
Dec. 2003 |
| #11 |
Shanghai
International Industry Fair (SIF), Shanghai, P.R. China
Nov. 2003 |
| #10 |
Energy
Asia 2003
PTC Asia 2003
CeMAT Asia 2003
Factory Automation Asia 2003
Shanghai, P.R. China
Nov. 2003 |
| #9 |
2003
WATER KOREA
Nov. 2003 |
| #8 |
NESC
2003 - 6th Int'l Conference on New Energy Systems & Conversions
Nov. 2003 |
| #7 |
Impressions
from Busan
South-Korea
Nov. 2003 |
| #6 |
2003
Fuel Cell Seminar
Miami Beach, Florida, USA
Nov. 2003 |
| #5 |
Impressions
from Shanghai, Beijing, P.R. China
Nov 2003 - Jul. 2004 |
| #4 |
f-cell
forum, Stuttgart, Germany
Sep. 2003 |
| #3 |
Hypothesis
V, Porto Conte, Italy
Sep. 2003 |
| #2 |
1st
European Hydrogen Energy Conference, Grenoble, France
Sep. 2003 |
| #1 |
Cooperation
for Energy Independence of Democracies in the 21st Century
Jerusalem, Israel
Aug. 2003 |
|
 |
 |
 |
 |
Shanghai International
Industry Fair (SIF) 2004
|
 |
|
 |
| Corporate
Information |
| home |
|
 |
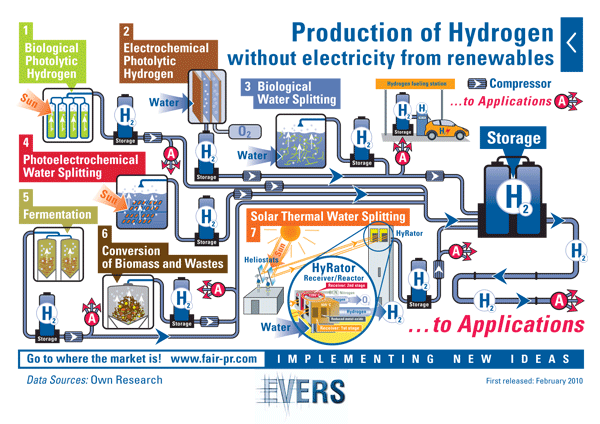
Production of Hydrogen without electricity from
renewables
Although a renewable energy source in conjunction with electrolysis would eliminate the dependence on fossil fuels, it still requires the production of electricity in the first place. The overall efficiencies of these processes are thereby reduced. Alternative methods without the need for electrical power include:
Photoelectrochemical (PEC) hydrogen
PEC systems use sunlight directly to generate sufficient energy to split water into hydrogen and oxygen. The advantage over conventional electrolysis using photovoltaic is the elimination of an electrical current network and the associated current transmission losses.
Biological Photolytic Hydrogen
Another way to directly tap solar energy for hydrogen production is to take advantage of certain microalgae and photosynthetic bacteria that sometimes use photosynthesis to make hydrogen instead of sugar and oxygen. However, the algal enzymes that trigger hydrogen production are inhibited by oxygen, so bioengineering of enzymes or a whole new organism would be required to make this process even remotely practical.
Conversion of Biomass and Wastes
Hydrogen can be produced via pyrolysis (thermochemical conversion) or anaerobic# digestion (fermentation) of biomass resources such as agricultural residues, wastes including plastics and waste grease; or biomass specifically grown for energy uses. Specific research areas include reforming of pyrolysis streams and development and testing of fluidizable catalysts.
Scientists are also working on “dark fermentation” reactions which do not do not require light energy at all. Here, a variety of bacteria ferment sugars and produce hydrogen using multi-enzyme systems. Sugars are relatively expensive substrates so engineering pretreatment technologies to convert lignocelluloses biomass into sugar-rich feedstock including hemicelluloses and cellulose that can be fermented directly to produce hydrogen, ethanol, and other high-value chemicals will be needed.
Solar Thermal Water Splitting
Water usually decomposes at temperatures of more than 2,500°C into hydrogen and oxygen. Researchers have demonstrated that highly concentrated sunlight can be used to generate these temperatures. However, catalysts based on metals or inorganic sulfur compounds can lower the heat needed to the more moderate range of 800 – 1,200°C. Such high-temperature, high-flux solar driven thermo-chemical processes offer a novel approach for the environmentally benign production of hydrogen, and is explored in more detail here:
https://www.hydrogenambassadors.com/background/direct-hydrogen-production-from-solar-energy.php
All the above mentioned methods are still in experimental phases and capable of supplying only small amounts of hydrogen. It seems that many technical, economical, and even mental hurdles need to be overcome before widespread commercial scale production of clean hydrogen is possible.
Download pdf
|
 |
|




